Planck's equation
Planck's equation describes the amount of spectral radiance at a certain wavelength radiated by a black body in thermal equilibrium.
Electromagnetics
editIn terms of wavelength (λ), Planck's [equation] is written [as]
where B is the spectral radiance, T is the absolute temperature of the black body, kB is the Boltzmann constant, h is the Planck constant, and c is the speed of light.
This form of the equation contains several constants that are usually not subject to variation with wavelength. These are h, c, and kB. They may be represented by simple coefficients: c1 = 2h c2 and c2 = h c/kB.
By setting the first partial derivative of Planck's equation in wavelength form equal to zero, iterative calculations may be used to find pairs of (λ,T) that to some significant digits represent the peak wavelength for a given temperature and vice versa.
Or,
Use c2 = 1.438833 cm K.
Problem 1
editFor the wavelength, temperature pair (570 nm, 5260 K), and for a simple calculator,
y=(1.48833/(0.0000570*5260))*exp(1.48833/(0.0000570*5260))/(exp(1.48833/(0.0000570*5260))-1)-5, followed by print y, yields a value close to zero (-1.006663E-03). The closer to zero the more accurate the estimate.
| Color | Frequency | Wavelength |
|---|---|---|
| violet | 668–789 THz | 380–450 nm |
| blue | 631–668 THz | 450–475 nm |
| cyan | 606–630 THz | 476–495 nm |
| green | 526–606 THz | 495–570 nm |
| yellow | 508–526 THz | 570–590 nm |
| orange | 484–508 THz | 590–620 nm |
| red | 400–484 THz | 620–750 nm |
For each color band, pick a wavelength, and calculate the corresponding temperature to complete the pair.
Problem 2
editThe table below gives approximate ultraviolet wavelength bands. Pick a wavelength from each and calculate the corresponding temperature to complete the pair.
| Name | Abbreviation | Wavelength range in nanometers | Energy per photon |
|---|---|---|---|
| Before UV spectrum | Visible light | above 400 nm | below 3.10 eV |
| Ultraviolet A, long wave, or black light | UVA | 400 nm–315 nm | 3.10–3.94 eV |
| Near | NUV | 400 nm–300 nm | 3.10–4.13 eV |
| Ultraviolet B or medium wave | UVB | 315 nm–280 nm | 3.94–4.43 eV |
| Middle | MUV | 300 nm–200 nm | 4.13–6.20 eV |
| Ultraviolet C, short wave, or germicidal | UVC | 280 nm–100 nm | 4.43–12.4 eV |
| Far | FUV | 200 nm–122 nm | 6.20–10.2 eV |
| Vacuum | VUV | 200 nm–100 nm | 6.20–12.4 eV |
| Low | LUV | 100 nm–88 nm | 12.4–14.1 eV |
| Super | SUV | 150 nm–10 nm | 8.28–124 eV |
| Extreme | EUV | 121 nm–10 nm | 10.2–124 eV |
| Beyond UV range | X-rays | below 10 nm | above 124 eV |
Problem 3
editX-rays span 3 decades in wavelength, frequency and energy. From 10 to 0.1 nanometers (nm) (about 0.12 to 12 keV) they are classified as soft X-rays, and from 0.1 nm to 0.01 nm (about 12 to 120 keV) as hard X-rays.
Super soft X-rays have energies in the 0.09 to 2.5 keV.
For each of the types of X-rays, pick a representative wavelength and calculate the temperature to complete the pair.
Gamma rays have wavelengths less than 10 picometers (less than the diameter of an atom).
1015 Hz = PHz petahertz
1018 Hz = EHz exahertz
1021 Hz = ZHz zettahertz
1024 Hz = YHz yottahertz
Convert these frequencies to their corresponding wavelengths and calculate the temperature for each to complete the pair.
Problem 4
editAstronomers often divide the infrared spectrum as follows:[1]
| Designation | Abbreviation | Wavelength |
|---|---|---|
| Near Infrared | NIR | (0.7–1) to 5 µm |
| Mid Infrared | MIR | 5 to (25–40) µm |
| Far Infrared | FIR | (25–40) to (200–350) µm. |
These are the approximate ranges for photon energies of the infrared bands:
| Division Name | Wavelength | Photon Energy |
| Near-infrared | 0.75-1.4 µm | 0.9-1.7 eV |
| Short-wavelength infrared | 1.4-3 µm | 0.4-0.9 eV |
| Mid-wavelength infrared | 3-8 µm | 150-400 meV |
| Long-wavelength infrared | 8–15 µm | 80-150 meV |
| Far infrared | 15 - 1,000 µm | 1.2-80 meV |
These are the main infrared atmospheric windows:
| Wavelength range (micrometres) |
Astronomical bands | Telescopes |
|---|---|---|
| 0.65 to 1.0 | R and I bands | All major optical telescopes |
| 1.1 to 1.4 | J band | Most major optical telescopes and most dedicated infrared telescopes |
| 1.5 to 1.8 | H band | Most major optical telescopes and most dedicated infrared telescopes |
| 2.0 to 2.4 | K band | Most major optical telescopes and most dedicated infrared telescopes |
| 3.0 to 4.0 | L band | Most dedicated infrared telescopes and some optical telescopes |
| 4.6 to 5.0 | M band | Most dedicated infrared telescopes and some optical telescopes |
| 7.5 to 14.5 | N band | Most dedicated infrared telescopes and some optical telescopes |
| 17 to 25 | Q band | Some dedicated infrared telescopes and some optical telescopes |
| 28 to 40 | Z band | Some dedicated infrared telescopes and some optical telescopes |
| 330 to 370 | Some dedicated infrared telescopes and some optical telescopes | |
| 450 | submillimeter | Submillimeter telescopes |
Commonly used sub-divisions are
| Division Name | Abbreviation | Wavelength | Characteristics |
| Near-infrared | NIR, IR-A DIN | 0.75-1.4 µm | Defined by the water absorption, and commonly used in fiber optic telecommunication because of low attenuation losses in the SiO2 glass (silica) medium. Image intensifiers are sensitive to this area of the spectrum. Examples include night vision devices such as night vision goggles. |
| Short-wavelength infrared | SWIR, IR-B DIN | 1.4-3 µm | Water absorption increases significantly at 1,450 nm. The 1,530 to 1,560 nm range is the dominant spectral region for long-distance telecommunications. |
| Mid-wavelength infrared | MWIR, IR-C DIN. Also called intermediate infrared (IIR) | 3-8 µm | In guided missile technology the 3-5 µm portion of this band is the atmospheric window in which the homing heads of passive IR 'heat seeking' missiles are designed to work, homing on to the Infrared signature of the target aircraft, typically the jet engine exhaust plume |
| Long-wavelength infrared | LWIR, IR-C DIN | 8–15 µm | This is the "thermal imaging" region, in which sensors can obtain a completely passive picture of the outside world based on thermal emissions only and requiring no external light or thermal source such as the sun, moon or infrared illuminator. Forward-looking infrared (FLIR) systems use this area of the spectrum. This region is also called the "thermal infrared." |
| Far infrared | FIR | 15 - 1,000 µm | (see also far-infrared laser). |
Pick a representative wavelength from each band and calculate its temperature to complete the pair.
Problem 5
edit“[T]erahertz radiation refers to electromagnetic waves propagating at frequencies in the terahertz range. It is synonymously termed submillimeter radiation, terahertz waves, terahertz light, T-rays, T-waves, T-light, T-lux, THz. The term typically applies to electromagnetic radiation with frequencies between high-frequency edge of the microwave band, 300 gigahertz (3 x 1011 Hz),"[2] and the long-wavelength edge of far-infrared light, 3000 GHz (3 x 1012 Hz or 3 THz). In wavelengths, this range corresponds to 0.1 mm (or 100 µm) infrared to 1.0 mm microwave.
Microwaves, a subset of radio waves, have wavelengths ranging from as long as one meter to as short as one millimeter, or equivalently, with frequencies between 300 MHz (0.3 GHz) and 300 GHz.[3] This broad definition includes both UHF and EHF (millimeter waves), and various sources use different boundaries.[4] In all cases, microwave includes the entire SHF band (3 to 30 GHz, or 10 to 1 cm) at minimum, with RF engineering often putting the lower boundary at 1 GHz (30 cm), and the upper around 100 GHz (3 mm).
Radio waves have frequencies from 300 [Gigahertz] GHz to as low as 3 [Kilohertz] kHz, and corresponding wavelengths from 1 millimeter to 100 kilometers.
Pick representative wavelengths from each band or sub-band and calculate a matching temperature to complete the pair.
Problem 6
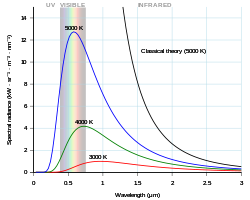
editThe first equation at top for the problem set allows the calculation of spectral radiance. Using a simple graphics routine calculate representative spectra for each wavelength, temperature pair.
Try integrating Planck's equation at the top of the resource to give the area under any such curve. Calculate the areas under each curve you've plotted. What is this area under the curve called?
Hypotheses
edit- Planck's equation can be derived without the use of quanta.
See also
edit- Angular momentum and energy
- Column densities
- Cosmic circuits
- Energy phantoms
- Furlongs per fortnight
- Radiation astronomy/Problem set
- Radiation dosage
- Radiation astromathematics problems
- Spectrographs
- Star jumping
- Synchrotron radiation problem set
- Telescopes and cameras
- Unknown coordinate systems
- Unusual units
- Vectors and coordinates
References
edit- ↑ IPAC Staff. "Near, Mid and Far-Infrared". NASA ipac. Retrieved 2007-04-04.
- ↑ Hankwang (October 31, 2008). Terahertz radiation. pp. 1. http://en.wikipedia.org/w/index.php?title=Terahertz_radiation&diff=248835142&oldid=248831987. Retrieved 2012-08-05.
- ↑ Pozar, David M. (1993). Microwave Engineering Addison-Wesley Publishing Company. ISBN 0-201-50418-9.
- ↑ http://www.google.com/search?hl=en&defl=en&q=define:microwave&ei=e6CMSsWUI5OHmQee2si1DQ&sa=X&oi=glossary_definition&ct=title
External links
edit- International Astronomical Union
- NASA/IPAC Extragalactic Database - NED
- NASA's National Space Science Data Center
- Office of Scientific & Technical Information
- The SAO/NASA Astrophysics Data System
- Scirus for scientific information only advanced search
- SDSS Quick Look tool: SkyServer
- SIMBAD Astronomical Database
- SIMBAD Web interface, Harvard alternate
- Spacecraft Query at NASA
- Universal coordinate converter
{{Charge ontology}}
{{Radiation astronomy resources}}{{Technology resources}}